|
3/17/2023 0 Comments Ionic radius vs atomic radiusThis question really is helpful in fusing general chemistry with cell biology, something you need to be able to do on the MCAT. This is why single ions can easily migrate through, but polyatomic ions cannot. Atomic radius decreases across a Period, from left to right, of the Periodic Table 2. The second part of this question is to get you to realize that protein channels are held together by hydrogen bonding, so they are rather narrow. Atomic radius 1 increases down a Group, from top to bottom, of the Periodic Table. Ionic radius is a measure of an atoms ion in a crystal lattice and which is a half distance between two ions that are barely touching each other. Ionic radius is a measure of an atoms ion in a crystal lattice and which is a half distance between two ions that are barely touching each other. Atomic radius is defined as a distance from the center of the nucleus to the outermost shell containing the electrons. As such, sodium cation is smaller than chloride anion. Atomic radius is defined as a distance from the center of the nucleus to the outermost shell containing the electrons. This causes the shell to swell slightly, resulting in a larger ion. When chlorine gains an election to become chloride, the valence shell has a greater negative charge, which results in repulsion. However, when sodium loses its valence electron from the third shell, it now has an an outer shell of n=2 coupled with a natural drop in size as the electron cloud loses and electron and does not repel as much. So neutral sodium has an atomic radii larger than neutral chlorine (about 151pm to 79 pm). If the atom loses an electron and becomes a cation,the atom becomes positively charged and the ionic radius is smaller than the atomic radius because the atom loses an electron energy shell. This isn't true for anions and cations, though. 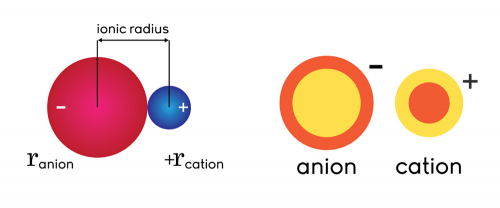
The radius of each atom in an ionic bond will be different due. When the atom is neutral, the atomic and ionic radius are the same.  
This is due to the increasing effective nuclear charge, which contracts the electron could. Ionic Radius : The ionic radius is the radius of an atom forming ionic bond or an ion. Sodium (element #11) and Chlorine (element #17) are in the same row of the periodic table, so as neutral elements they follow the size trend of descending radii moving left-to-right. On one level, you need to know that ionic radii vary from elemental radii. The average distance between the center of the nucleus and the electron cloud of outermost orbit is called atomic radius. The purpose of this question is two-fold. I assume you are referring to one of our (that being BR's) general chemistry questions here.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
